Assembly of recombinant tau into filaments identical to those of
Por um escritor misterioso
Last updated 22 dezembro 2024
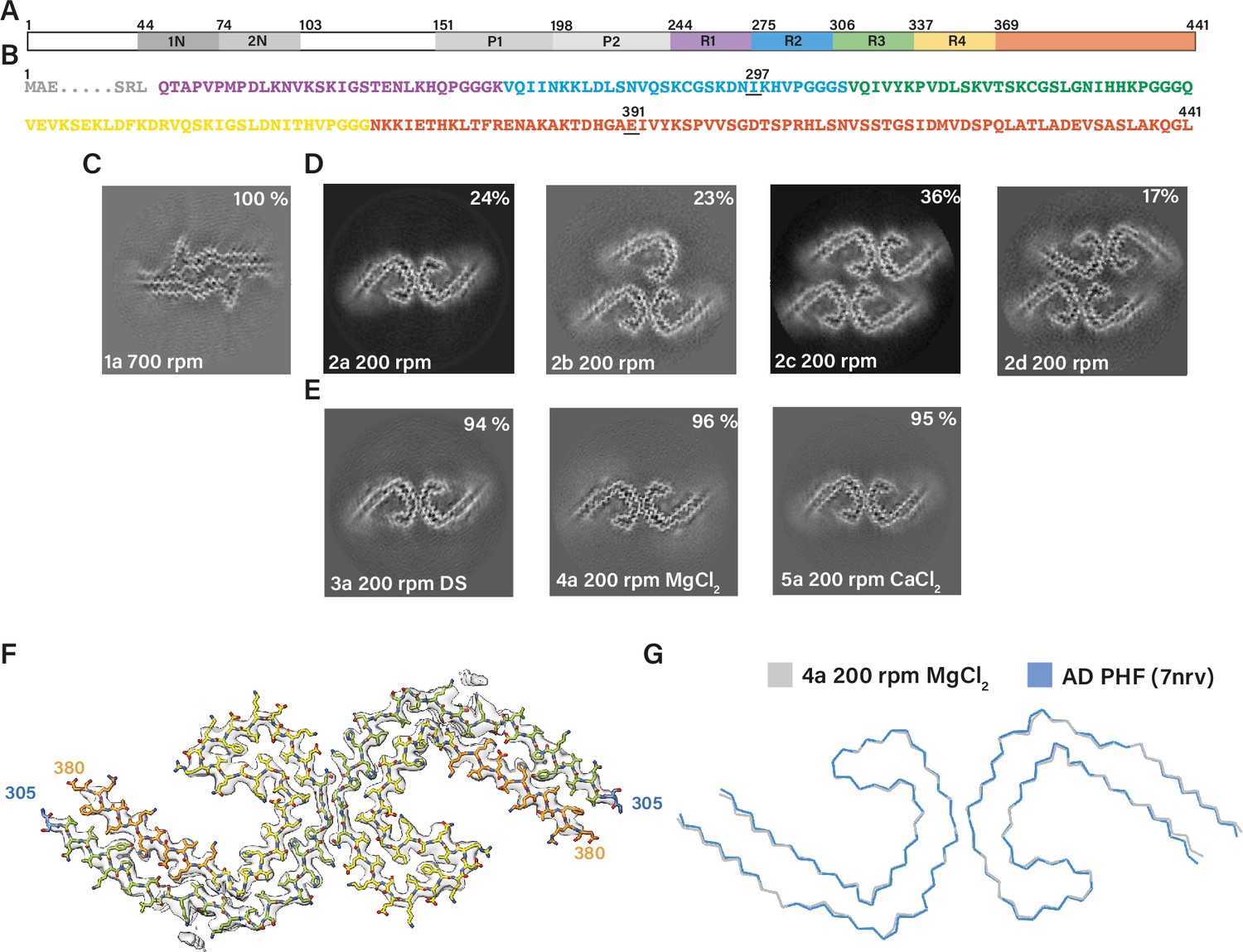
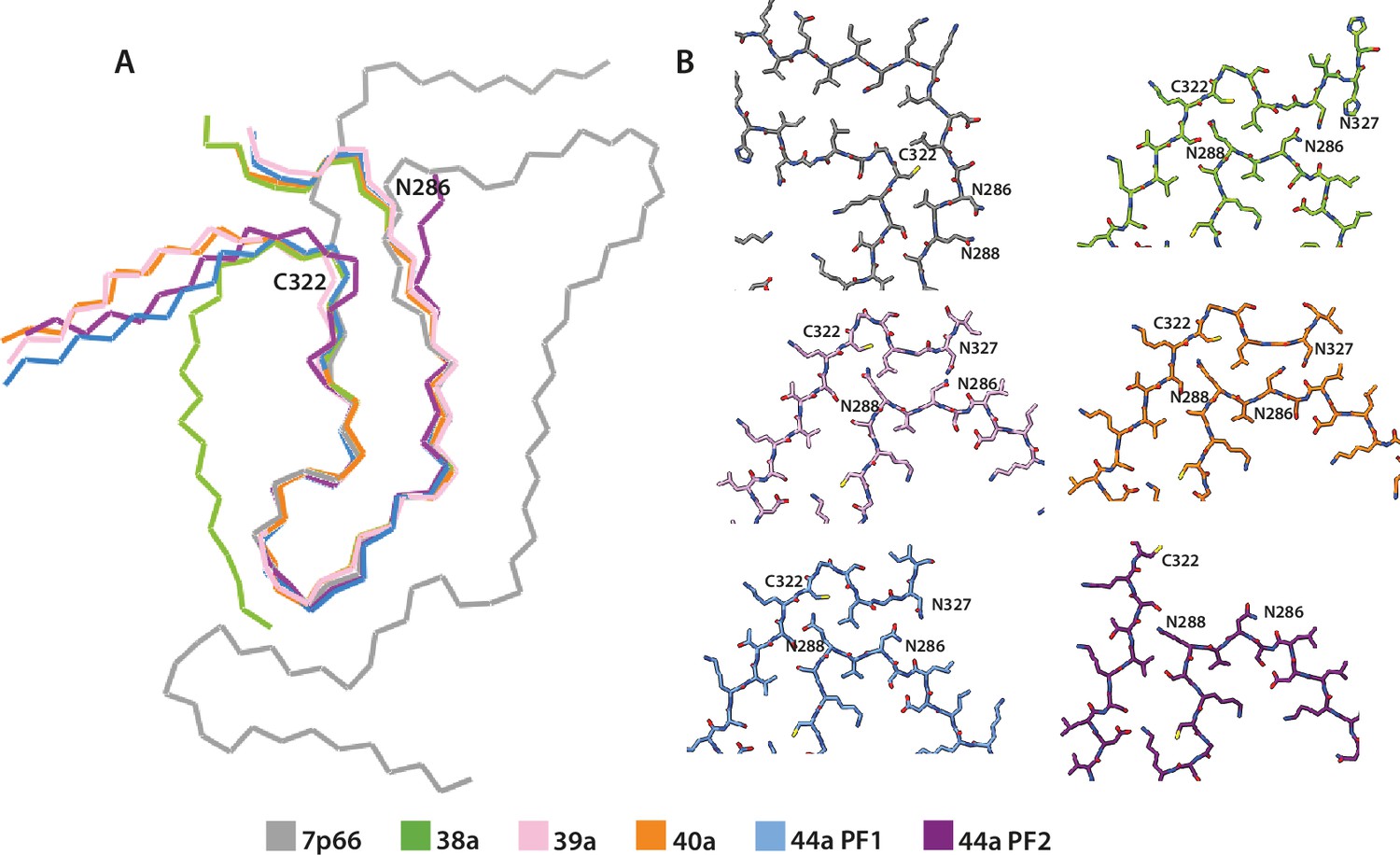
Many neurodegenerative diseases, including Alzheimer’s disease, the most common form of dementia, are characterised by knotted clumps of a protein called tau. In these diseases, tau misfolds, stacks together and forms abnormal filaments, which have a structured core and fuzzy coat. These sticky, misfolded proteins are thought to be toxic to brain cells, the loss of which ultimately causes problems with how people move, think, feel or behave. Reconstructing the shape of tau filaments using an atomic-level imaging technique called electron cryo-microscopy, or cryo-EM, researchers have found distinct types of tau filaments present in certain diseases. In Alzheimer’s disease, for example, a mixture of paired helical and straight filaments is found. Different tau filaments are seen again in chronic traumatic encephalopathy (CTE), a condition associated with repetitive brain trauma. It remains unclear, however, how tau folds into these distinct shapes and under what conditions it forms certain types of filaments. The role that distinct tau folds play in different diseases is also poorly understood. This is largely because researchers making tau proteins in the lab have yet to replicate the exact structure of tau filaments found in diseased brain tissue. Lövestam et al. describe the conditions for making tau filaments in the lab identical to those isolated from the brains of people who died from Alzheimer’s disease and CTE. Lövestam et al. instructed bacteria to make tau protein, optimised filament assembly conditions, including shaking time and speed, and found that bona fide filaments formed from shortened versions of tau. On cryo-EM imaging, the lab-produced filaments had the same left-handed twist and helical symmetry as filaments characteristic of Alzheimer’s disease. Adding salts, however, changed the shape of tau filaments. In the presence of sodium chloride, otherwise known as kitchen salt, tau formed filaments with a filled cavity at the core, identical to tau filaments observed in CTE. Again, this structure was confirmed on cryo-EM imaging. Being able to make tau filaments identical to those found in human tauopathies will allow scientists to study how these filaments form and elucidate what role they play in disease. Ultimately, a better understanding of tau filament formation could lead to improved diagnostics and treatments for neurodegenerative diseases involving tau.
Laboratory-based methods are presented that produce filamentous tau aggregates with the same structures as those observed in neurodegenerative disease.
Laboratory-based methods are presented that produce filamentous tau aggregates with the same structures as those observed in neurodegenerative disease.
Assembly of recombinant tau into filaments identical to those of Alzheimer's disease and chronic traumatic encephalopathy
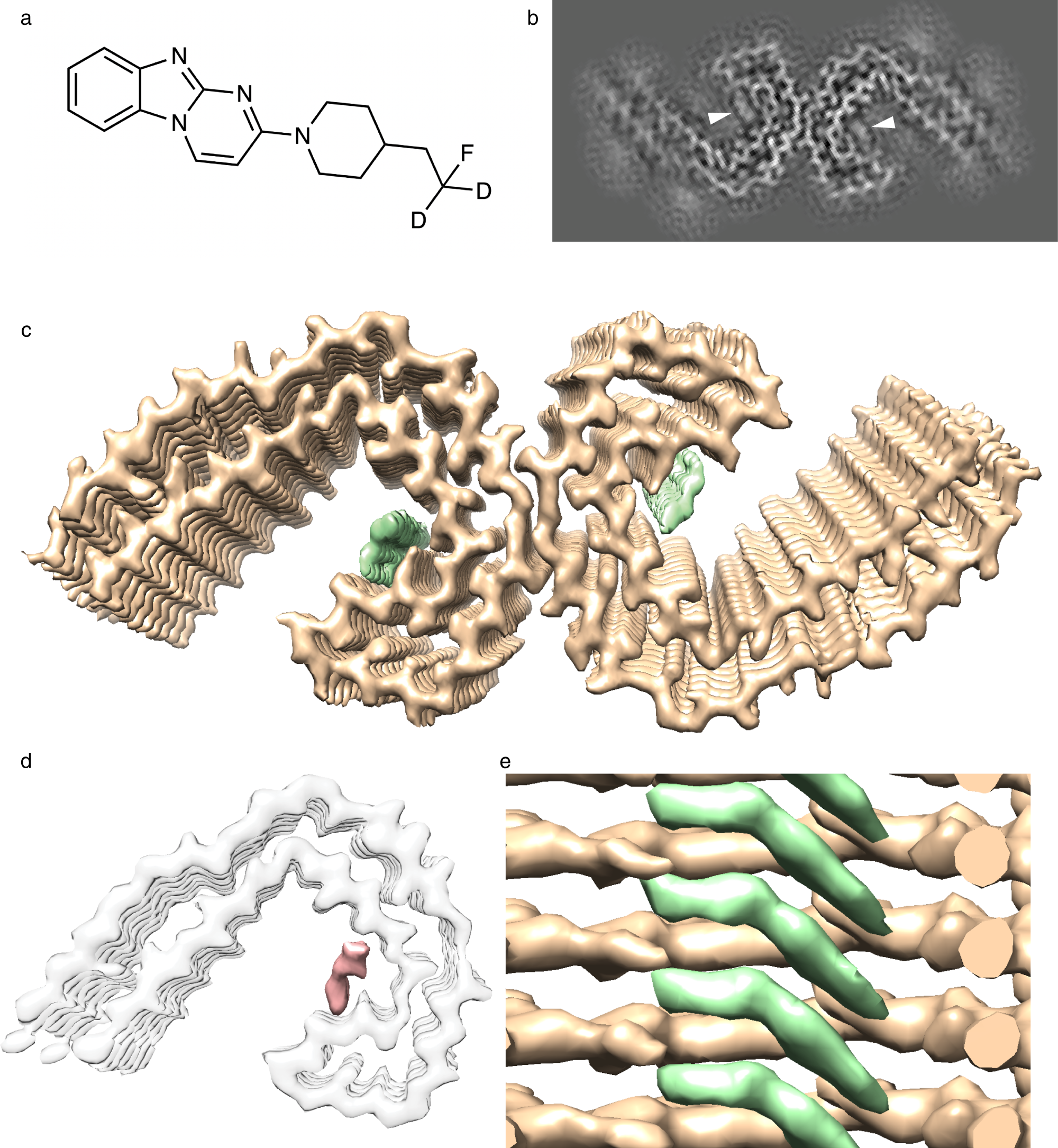
Stacked binding of a PET ligand to Alzheimer's tau paired helical filaments
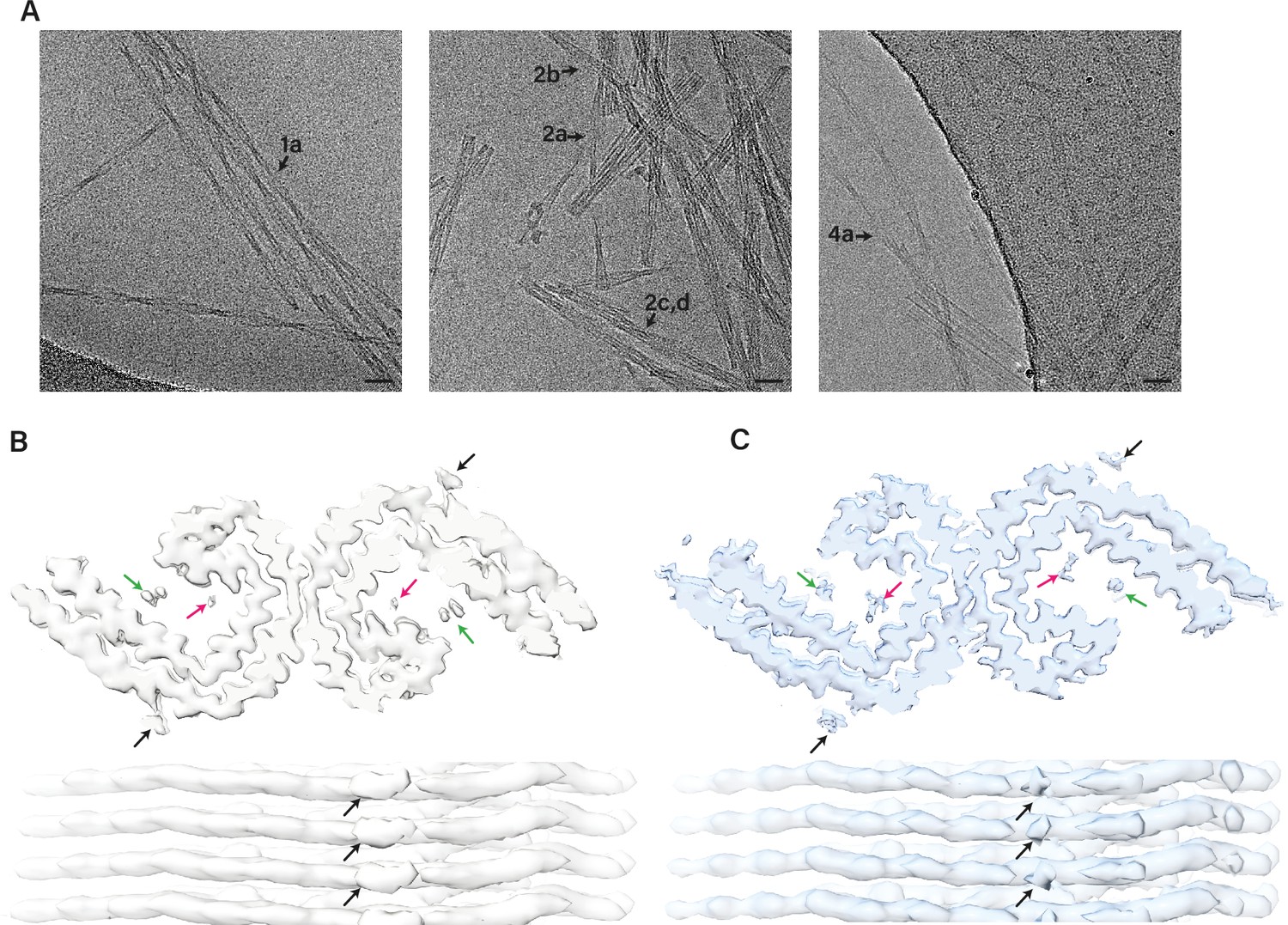
Assembly of recombinant tau into filaments identical to those of Alzheimer's disease and chronic traumatic encephalopathy
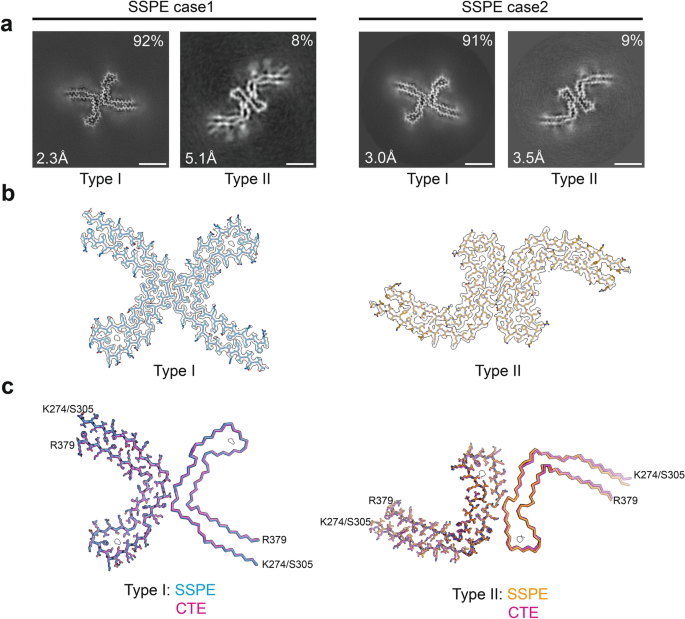
Identical tau filaments in subacute sclerosing panencephalitis and chronic traumatic encephalopathy, Acta Neuropathologica Communications

Cryo‐EM structures of tau filaments from SH‐SY5Y cells seeded with brain extracts from cases of Alzheimer's disease and corticobasal degeneration - Tarutani - 2023 - FEBS Open Bio - Wiley Online Library

Assembly of recombinant tau into filaments identical to those of Alzheimer's disease and chronic traumatic encephalopathy. - Abstract - Europe PMC
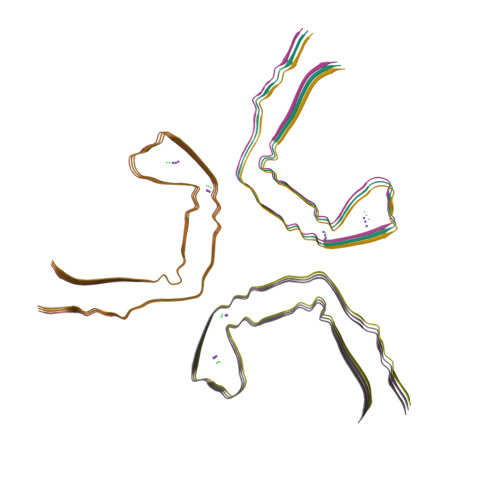
RCSB PDB - 7QK5: In vitro assembled 266/297 - 391 tau filaments with KCl (10a)
Assembly of recombinant tau into filaments identical to those of Alzheimer's disease and chronic traumatic encephalopathy
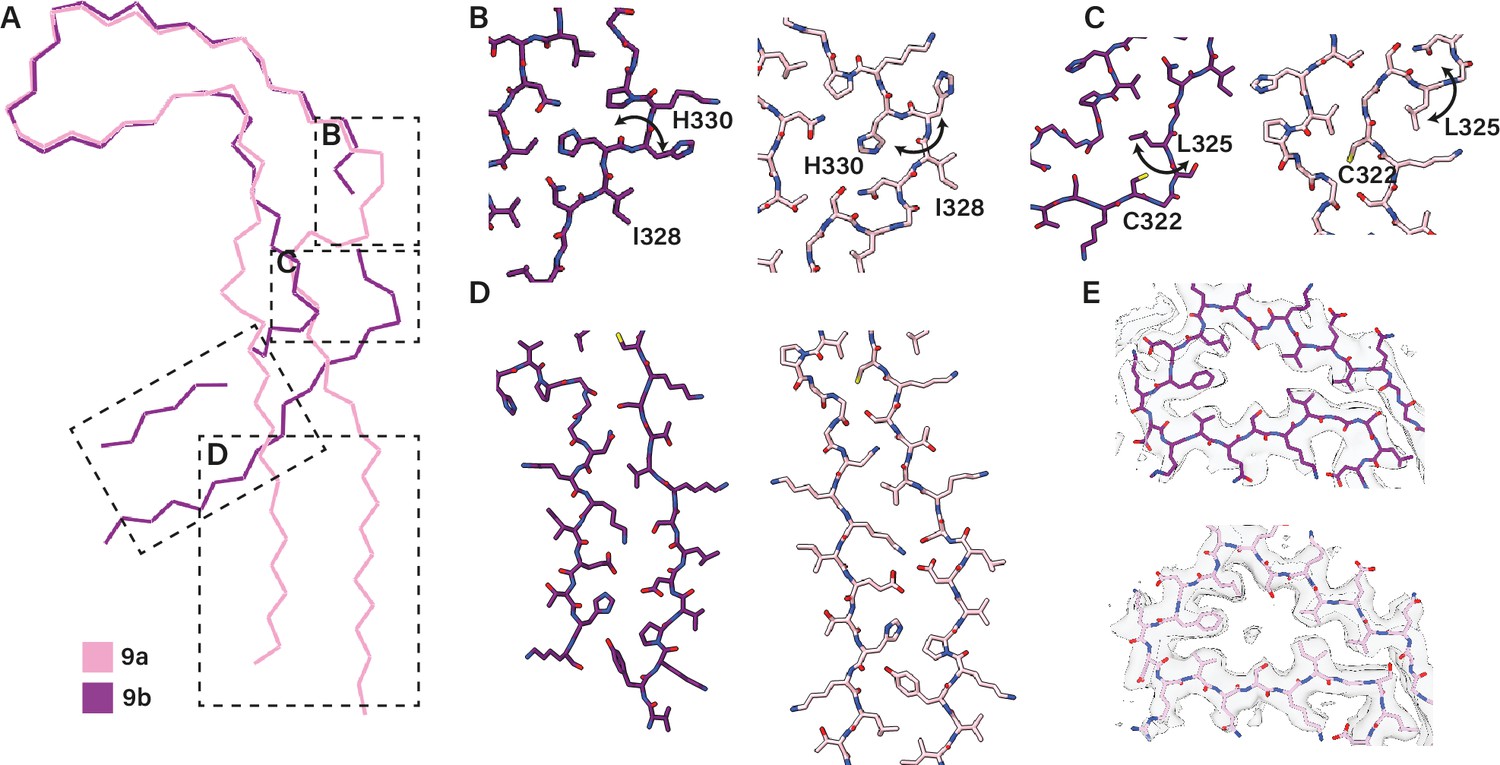
Assembly of recombinant tau into filaments identical to those of Alzheimer's disease and chronic traumatic encephalopathy
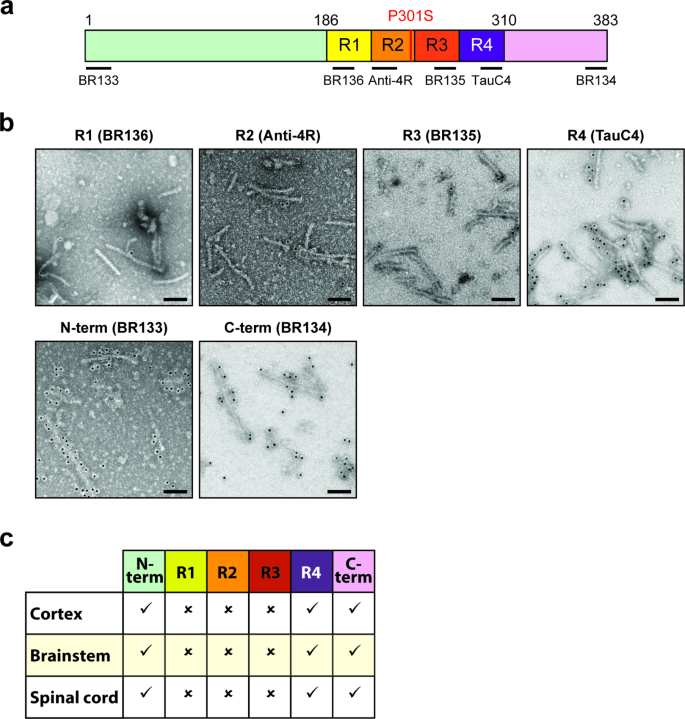
Cryo-EM structures of tau filaments from the brains of mice transgenic for human mutant P301S Tau, Acta Neuropathologica Communications
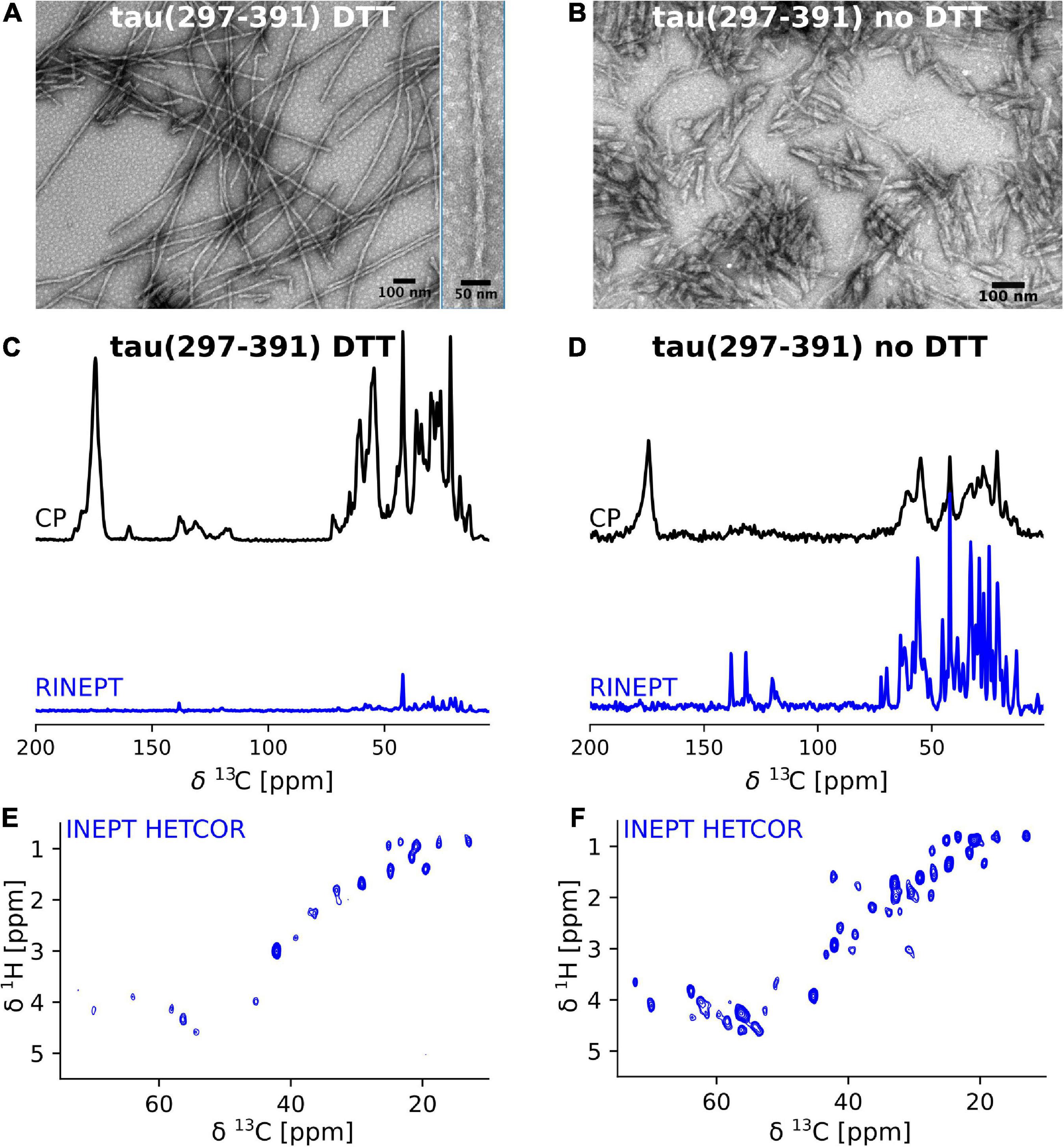
Frontiers Solid-state NMR of paired helical filaments formed by the core tau fragment tau(297-391)
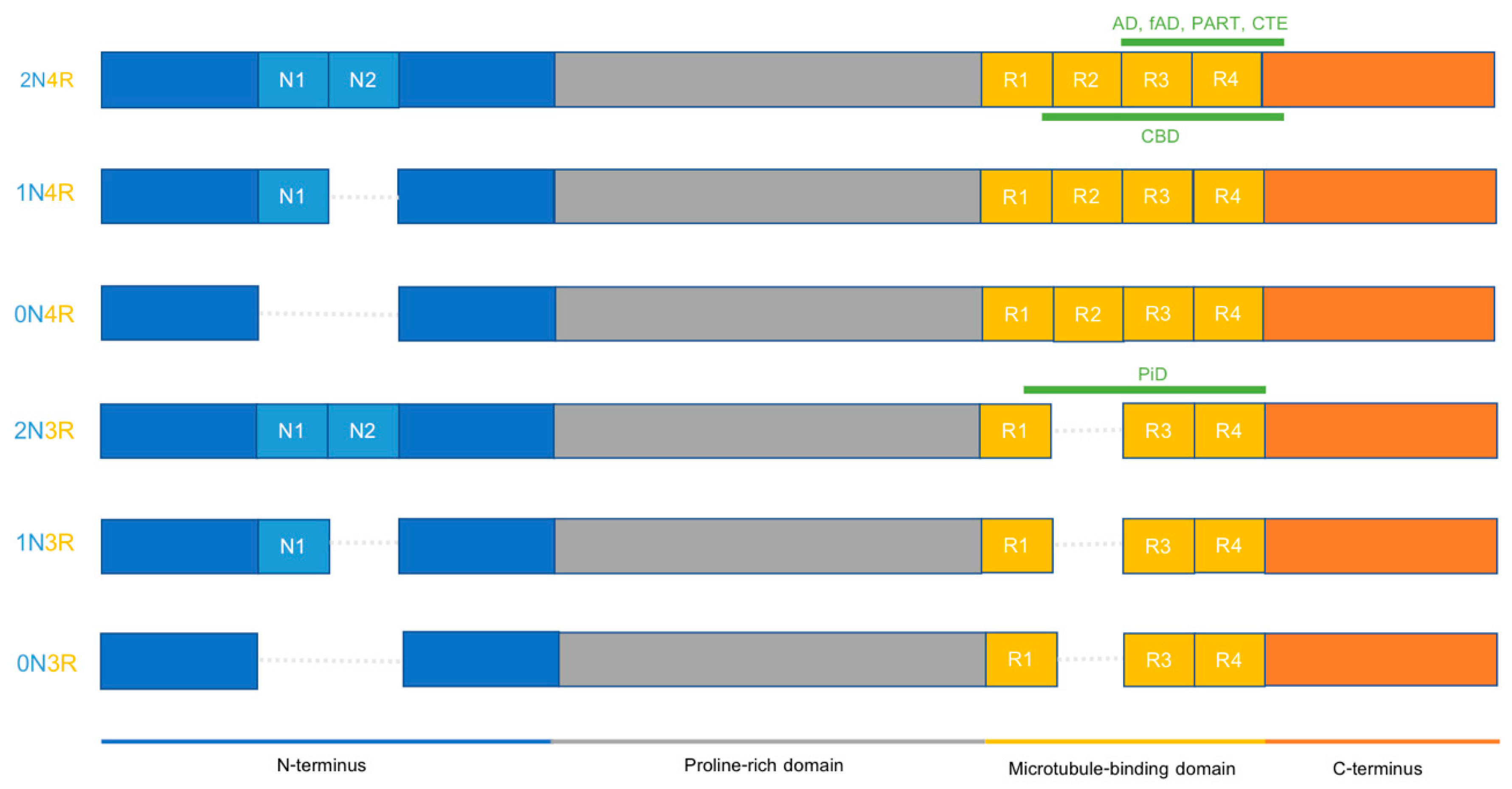
IJMS, Free Full-Text
Disease-specific tau filaments assemble via polymorphic intermediates

Cryo‐EM structures of tau filaments from SH‐SY5Y cells seeded with brain extracts from cases of Alzheimer's disease and corticobasal degeneration - Tarutani - 2023 - FEBS Open Bio - Wiley Online Library

RCSB PDB - 7QL3: in vitro assembled 266/297 - 391 tau filaments with NaCl (8b)
Recomendado para você
-
 Brain Test: Tricky Brain Test Riddles Game for Android - Download22 dezembro 2024
Brain Test: Tricky Brain Test Riddles Game for Android - Download22 dezembro 2024 -
 攻略・解答】Happy Glass【Level 291~Level 300】22 dezembro 2024
攻略・解答】Happy Glass【Level 291~Level 300】22 dezembro 2024 -
 Mindful Leadership Stock Illustrations – 297 Mindful Leadership22 dezembro 2024
Mindful Leadership Stock Illustrations – 297 Mindful Leadership22 dezembro 2024 -
 Ep. #297. Flat Earthers the true test of your ability to engage22 dezembro 2024
Ep. #297. Flat Earthers the true test of your ability to engage22 dezembro 2024 -
 5th Grade Fall Math Logic Puzzles Activities22 dezembro 2024
5th Grade Fall Math Logic Puzzles Activities22 dezembro 2024 -
 Grace Hudson on X: A spot of osmosis to brighten your Wednesday22 dezembro 2024
Grace Hudson on X: A spot of osmosis to brighten your Wednesday22 dezembro 2024 -
 Analysis of Characteristics of Patients with Abdominal Pain in the22 dezembro 2024
Analysis of Characteristics of Patients with Abdominal Pain in the22 dezembro 2024 -
Solved Using the dataset for the assignment, run a22 dezembro 2024
-
 Draw Toilet : Squid Game APK for Android Download22 dezembro 2024
Draw Toilet : Squid Game APK for Android Download22 dezembro 2024 -
 Brain Scans Reveal Dogs' Thoughts22 dezembro 2024
Brain Scans Reveal Dogs' Thoughts22 dezembro 2024
você pode gostar
-
 How can Google make a mistake like this??? - Google Search Central Community22 dezembro 2024
How can Google make a mistake like this??? - Google Search Central Community22 dezembro 2024 -
 GTA 5 PC Mods - Online Garage in Story Mode with Editor22 dezembro 2024
GTA 5 PC Mods - Online Garage in Story Mode with Editor22 dezembro 2024 -
Do y'all want this one?, songs with lyrics22 dezembro 2024
-
 Figure (doors-roblox)22 dezembro 2024
Figure (doors-roblox)22 dezembro 2024 -
 Kit Volante Logitech G29 Driving Force + Headset astro Gaming A10 - PS5, PS4, PS3 e pc em Promoção na Americanas22 dezembro 2024
Kit Volante Logitech G29 Driving Force + Headset astro Gaming A10 - PS5, PS4, PS3 e pc em Promoção na Americanas22 dezembro 2024 -
𝑶 𝒗𝒂𝒍𝒐𝒓 𝒅𝒂 𝒗𝒊𝒅𝒂 𝒆𝒔𝒕𝒂́ 𝒏𝒂𝒔 𝒑𝒆𝒒𝒖𝒆𝒏𝒂𝒔 𝒄𝒐𝒊𝒔22 dezembro 2024
-
 The only thing scarier than Herobrine is a Herobrine Girl ---- no, what's scary is when they both come at you with diamo… en 202322 dezembro 2024
The only thing scarier than Herobrine is a Herobrine Girl ---- no, what's scary is when they both come at you with diamo… en 202322 dezembro 2024 -
Buu VS The Multiverse - Chapter 88, Page 2052 - DBMultiverse22 dezembro 2024
-
 Dommaraju Gukesh reaches the 11th spot in the live World rankings22 dezembro 2024
Dommaraju Gukesh reaches the 11th spot in the live World rankings22 dezembro 2024 -
 Its crazy how much stand models differ between Jojo roblox games22 dezembro 2024
Its crazy how much stand models differ between Jojo roblox games22 dezembro 2024